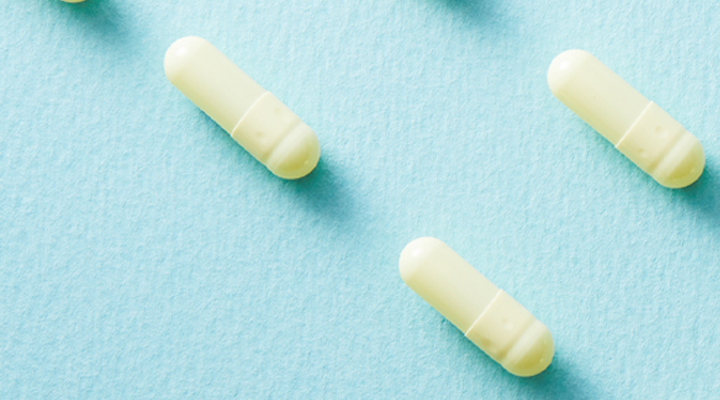
[hy-1 발효홍삼] 논문 _ Enhanced Absorption Study of Ginsenoside Compound K (20-O-β-(D-Glucopyranosyl)-20(S)-protopanaxadiol) after Oral Administration of Fermented Red Ginseng Extract (HYFRG™) in Healthy Korean Volunteers and Rats
2021-06-04